| |
| Int J Tuberc Lung Dis. 2009 November; 13(11): 1433–9. Tobacco use and its determinants in HIV-infected patients on antiretroviral therapy in West African countries Antoine Jaquet,1* Didier-Koumavi Ekouevi,1,2 Maiga Aboubakrine,3 Jules Bashi,4 Eugène Messou,5 Moussa Maiga,3 Hamar-Alassane Traore,6 Marcel Zannou,4 Calixte Guehi,7 Franck-Olivier Ba-Gomis,8 Albert Minga,9 Gérard Allou,2 Serge-Paul Eholie,10 Francois Dabis,1 Emmanuel Bissagnene,10 and Annie-Jeanne Sasco1 1Centre épidémiologie et biostatistique
INSERM : U897, Université Victor Segalen - Bordeaux II, FR 2Programme PAC-CI
CHU de Treichville, Abidjan,CI 3Service d'Hépato-Gastro-Entérologie
Hôpital Gabriel Touré, Bamako,ML 4Centre de Prise en Charge des Personnes vivant avec le VIH
CHNU, Cotonou,BJ 5Centre de Prise en Charge de Recherches et de Formation
ACONDA-CePReF, Adultes, Abidjan,CI 6Centre de Prise en Charge des Personnes vivant avec le VIH
Hôpital du point G, Bamako,ML 7Unité de Soins Ambulatoires et de Conseil
(USAC), Abidjan,CI 8Centre Intégré de Recherche Bioclinique d'Abidjan
(CIRBA), Abidjan,CI 9Centre Médical de Suivi de Donneurs de Sang
CNTS/PRIMO-CI, Abidjan,CI 10SMIT, Service de Maladies Infectieuses et Tropicales
Hôpital de Treichville, Abidjan,CI |
INTRODUCTION Tobacco smoking is common in HIV-infected patients from industrialized countries. In West Africa, few data exist concerning tobacco consumption. METHODS A cross-sectional survey was conducted within the International epidemiological Database to Evaluate AIDS (IeDEA) network in West Africa. Health workers administered to patients receiving antiretroviral treatment a questionnaire assessing tobacco and cannabis consumption. Regular smokers were defined as present smokers who smoked >1 cigarette per day for ≥1 year. RESULTS Overall, 2920 patients were enrolled in three countries. The prevalence of ever smokers and present smokers were 46.2% (95% CI 42.8–49.5) and 15.6% (95% CI 13.2–18.0) in men and 3.7% (95% CI 2.9–4.5) and 0.6% (95% CI 0.3–0.9) in women, respectively. Regular smoking was associated being from Côte d’Ivoire or Mali compared to Benin (OR 4.6; 95% CI 2.9–7.3 and 7.7; 95% CI 4.4–13.6), a severely impaired immunological status at HAART initiation (OR 1.5; 95% CI 1.1–2.2) and a history of tuberculosis (OR 1.8; 95% CI 1.1–3.0). CONCLUSION Marked differences of smoking prevalence exist between these West African countries. This survey approach also provides evidences concerning the association between cigarette smoking and tuberculosis in HIV-infected patients, a major public health issue in this part of the world. MESH keywords: Adult, Anti-Retroviral Agents, therapeutic use, Antiretroviral Therapy, Highly Active, Benin, epidemiology, Cote d'Ivoire, epidemiology, Cross-Sectional Studies, Databases as Topic, Female, HIV Infections, drug therapy, epidemiology, Humans, Logistic Models, Male, Mali, epidemiology, Marijuana Smoking, epidemiology, Middle Aged, Odds Ratio, Prevalence, Questionnaires, Smoking, epidemiology, Tuberculosis, epidemiology Author keywords: HIV/AIDS, tobacco, tuberculosis, prevalence estimate, sub-Saharan Africa |
Since the introduction of Highly Active Antiretroviral therapy (HAART), the pattern of AIDS-defining and non-AIDS defining disease conditions has evolved. Chronic manifestations such as cancers or cardiovascular diseases are becoming an emerging problem among HIV-infected persons in industrialized countries [1]. Questions have been raised concerning the relative contribution of well characterized risk factors of non-AIDS related cancers such as tobacco consumption. Indeed, the prevalence of smoked tobacco has been shown to be particularly high in HIV-infected patients living in industrialized countries, ranging from 40% to 70% according to studies from European and North American HIV-infected cohorts [2–5], and two to three times higher than in the corresponding general population [2, 6, 7]. In sub-Saharan Africa, few data exist concerning tobacco consumption particularly in West African countries. Since this well known cancer risk factor is a growing problem in this part of the world and is amenable to prevention strategies, there is a particular need to document its current level in this specific population. We thus aimed at estimating the prevalence of tobacco and cannabis consumption among West African HIV-infected subjects treated with HAART. |
Study population This cross-sectional survey was conducted within the International epidemiological Database to Evaluate AIDS (IeDEA) network ( http://www.iedea-hiv.org). By collecting and harmonizing data from multiple HIV/AIDS cohorts from industrialized and resource-limited countries, this initiative aims to address unique and evolving research questions in the field of HIV/AIDS care and treatment. In the West African region, this collaboration was initiated in July 2006 and currently involves 14 adult HIV/AIDS clinics spread over eight countries: Benin, Burkina-Faso, Côte d’Ivoire, Gambia, Ghana, Mali, Nigeria and Senegal ( http://www.iedeawestafrica.org). Of these countries, three of them volunteered and had the logistic capacity to perform such a survey in a total of eight HIV treatment centers. The study population was assembled by enrolling consecutively all HIV-infected patients on treatment attending for consultation at the participating HIV clinics during a four-week period. For clinics that were unable to guarantee the exhaustivity in the enrollment procedure, patients were randomly selected. Systematic sampling, a form of one-stage cluster sampling, was used. Social health workers in charge of inclusions selected every n th HIV-infected patient on HAART (depending on the monthly number of attendees) after randomly selecting the first patient through n th statistical elements as the starting point. Patients included fulfilled the following criteria: HIV-1 or HIV-2 or HIV1+2 infected, on HAART defined as receiving three antiretroviral drugs for a minimum of one month, aged 18 or more and willing to participate to the survey. Study procedures Especially trained social health workers, administered during face-to-face interviews a standardized form assessing socio-demographic characteristics, tobacco and cannabis consumption. Data collected during these interviews were secondarily linked with the IeDEA clinical database, using the identification number attributed to each patient, in order to dispose of additional information that could not be collected during the interviews. This linkage allowed the use of baseline and follow-up clinical and biological data such as CD4 cell count and duration of HAART. A written informed consent was obtained from all study participants. This study was approved by national ethic committees from Côte d’Ivoire, Mali and Benin. Measures Questions related to present and past tobacco and cannabis consumption were assessed with a standardized questionnaire adapted from several forms previously used in epidemiological studies conducted in Africa [ 8, 9]. Regular smokers were defined as present smokers who smoked at least one cigarette per day for one year or more in accordance to the World Health Organization definition (WHO) [ 10]. Statistical analysis Analyses were first conducted to compare tobacco consumption characteristics according to countries using Pearson’s χ 2 test or Fisher’s exact test when appropriate for qualitative variables and t-test or Kruskall-Wallis test for quantitative variables. A logistic regression model was used for univariate and multivariate analyses of the determinants of smoking consumption. A stepwise descending procedure was applied to select variables that were significantly associated, using a P-value threshold of 0.20. Variables that were also identified in the literature as related to smoking tobacco were systematically included in the model. Proportion and Odds Ratio (OR) estimates were reported with their 95% Confidence Interval (95% CI). All statistical analyses were performed using SAS software, 9.1 (SAS Institute Inc, NC, USA). |
General characteristics A total of 2920 HAART-treated patients (1838 from five clinics in Côte d’Ivoire, 486 from one clinic in Benin and 596 from two clinics in Mali) were included in the present study. The male to female ratio from our study sample (0.41:1) did not differ significantly from the remaining 2380 HAART-treated HIV-infected patients seen in the participating clinical centers during the same study period and that could not be interviewed (0.42:1) (P=0.93). The median age of these 2920 patients was 38 years (interquartile range [IQR] 32–45 years) and the median duration on HAART was 3 years (IQR 1–4 years). The median CD4 count at HAART initiation was 162 cells/mm3 (IQR 76–245). The place of living was essentially urban for 90.8% of patients, 9.8% living in a rural area. Smoking prevalence Among the 2920 enrolled patients, the prevalence of present or past history and regular smoking consumption was 46.2% (95% CI 42.8–49.5) and 15.6% (95% CI 13.2–18.0) in men and 3.7% (95% CI 2.9–4.5) and 0.6% (95% CI 0.3–0.9) in women, respectively. In men, the prevalence of regular smokers in Mali (29.7% (95% CI 22.9–36.5)) was twice higher than in Côte d’Ivoire (14.2% (95% CI 11.2–17.2)) and 10 times higher than in Benin (2.9% (95% CI 0.4–5.4%)) ( figure 1). Irrespective of the country, the prevalence of women who smoked regularly was low, ranging from 0% in Benin to 1.4% (95% CI 0.3–2.6) in Mali. Characteristics of smoking consumption Exposed patients stating a present or former smoking consumption declared smoking a median number of 8 cigarettes per day, with a significant difference according to country. Malian smokers declared a median number of 10 cigarettes per day while smokers from Benin declared a median number of three cigarettes per day (p<0.0001). The proportion of women who declared a present or past history of smoking was significantly higher in Côte d’Ivoire compared to Benin or Mali (p=0.03). Almost all smokers declared buying manufactured and filtered cigarettes ( table 1). Factors associated with tobacco smoking Being a man, reporting alcohol use during the past year and being from Mali or Côte d’Ivoire compared to Benin were the main determinants of having a present or past history of regular tobacco smoking. Patients stating being single, widowed or divorced and older patients were also more likely to be regular smokers compared to married or cohabitants and younger patients. Patients with a severely impaired immunological status at HAART initiation and patients with a known history of tuberculosis were also more likely to have a present or past history of regular smoking ( table 2). Cannabis consumption Overall, the prevalence of present or past cannabis use was 1.7% (95% CI 1.3–2.2). This consumption, illicit in the three countries surveyed, was significantly more frequently reported by men (5%) compared to women (0.4%) (p<0.0001). The proportions of present or past cannabis consumers were 1.8% in Côte d’Ivoire, 1.7% in Mali and 1.5 % in Benin (p=0.82). |
Smoking prevalence The main finding of this survey is the country variability in the prevalence of smokers among this HIV-infected population. Smoking prevalence in HIV-infected patients from Mali was two-times higher than in Côte d’Ivoire and 10-times higher than in Benin and close to the estimates reported in industrialized countries. Little is known about smoking consumption in the general population from West African countries, especially French-speaking ones. In its third edition, “the tobacco Atlas” reports tobacco smoking prevalence estimates according to countries [ 11]. In Côte d’Ivoire, smoking prevalence estimates according to gender were 11.2% in men and 0.7% in women. A report from Koffi et al conducted in 180 patients recruited from the three teaching hospitals of Abidjan, Côte d’Ivoire found an overall prevalence of present or past smoking and present smoking of 31.7% and 16.1%, respectively [ 12]. A recent report published by Sodjinou et al assessed the prevalence of cardio-metabolic risk factors and their association with socio-economic factors in a randomly selected population of 200 adults in Cotonou, Benin. The estimate prevalence of present or past smoking and present smoking were 17% and 5% in men and 1% and 0% in women, respectively [ 13]. These previous findings from the adult population of Côte d’Ivoire and Benin, irrespective of their HIV status, suggest that smoking prevalence is quite comparable between HIV-infected and HIV-uninfected persons. In Mali, the current smoking prevalence estimate in the general adult population is 14% in men and 0.6% in women according to the tobacco atlas [ 11]. These results are about twice lower than the estimated prevalence in our survey suggesting a potential association between smoking and HIV status. While the few available results from the international studies on smoking prevalence in the general population place sub-Saharan Africa and particularly West African countries at a relatively low level of smoking consumption [14], our study highlights that smoking habits in HIV-infected patients and probably in the general population are not homogenous in this part of the world. We thus make a plea for extending those prevalence studies in other HIV-infected populations as well as in non HIV-infected populations from African countries. Tobacco consumption in HIV/AIDS patients from industrialized countries has been shown to be elevated and higher than in the general population with approximately half of patients being regular smokers. This high frequency of cigarettes smoking is at least partly related to the presence of a specific subpopulation of multidrug users which have a particularly high rate of smoking [2]. The lower prevalence of smokers found in HIV-infected cohorts from West African countries could be partly explained by the fact that this population does not share the same addiction characteristics. However, the proportion of smokers in HIV-infected patients from West Africa might grow in the coming years. Recent reports emphasized that tobacco industries are entering actively the market of low-resource countries, including Africa, in order to compensate for their losses in the Northern Hemisphere at a time when the demand in these countries is decreasing [15]. For this purpose, tobacco industry uses all kinds of strategies to market their products in these countries and faces practically no barriers to their business as many of West African countries do not have any policy to restrain tobacco advertising or other promotion strategies [16, 17]. Factors associated with smoking The important variation of smoking prevalence according to gender is in accordance with most previous studies conducted in HIV-negative populations on the African continent [ 14, 18]. There is some evidence that this particularly unbalanced sex ratio in smokers might now be evolving in West Africa. Reports from the “Global youth tobacco surveillance” showed that the prevalence figures of current smoking in randomly selected samples of schoolchildren in Benin, Côte d’Ivoire and Mali were 11.2% (95% CI 7.4–16.5), 19.3% (95% CI 16.1–23.0) and 41.8% (95% CI 34.0–50.0) in boys compared to 1.8% (95% CI 0.9–3.6), 7.1% (95% CI 5.1–9.9) and 4.6% (95% CI 2.7–7.7) in girls, respectively [ 19]. Although no reliable comparisons to our study population can be drawn from these results, these population-based prevalence estimates already showed higher figures for girls. A report from Simen-Kapeu et al found a smoking prevalence of 1.4% in 2067 HIV-infected women recruited in three HIV clinics in Côte d’Ivoire 10 years ago [ 20]. Although our study population was not recruited in the same HIV clinics in Abidjan, this reported prevalence measured 10 years ago on untreated populations [ 21] is more than twice lower than the 3.7% prevalence found in the 1331 HIV-infected women from Côte d’Ivoire enrolled in our present survey. The association found between tobacco use and a severely impaired immunological status (defined by having a CD4 cell count ≤50 cells per mm3) at HAART initiation has already been reported. The French Aquitaine ANRS Co3 cohort reported an association between current smoking and uncontrolled HIV infection (defined differently than in African cohorts as patients presenting with a CD4 cell count ≤350/mm3 or a plasma viral load ≥1000 copies/ml) with an OR of 1.24 (95% CI 1.02–1.50) [2]. Their authors have put forward that tobacco consumption could act as a comfort measure against the severity of HIV infection. This association between tobacco smoking and a worse immunological status at HAART initiation could also be related to the decrease in the immune response related to tobacco use [22]. Cigarette smoking has been associated with an increased risk of developing bacterial and viral pneumonia in both HIV-negative and HIV-positive patients [23–27]. The association between a low CD4 count at HAART initiation and cigarette smoking could be then related to their dual association with an increased risk of pulmonary infection. Indeed, the significant association between smoking and a history of tuberculosis infection is in accordance with this latter hypothesis. An association between tuberculosis and smoking has already been described in cross-sectional and case-control studies in low income countries such as India and Indonesia [28–30], but there is scarce information from sub-Saharan Africa where tuberculosis is highly prevalent. However, our study design did not allow us to draw any causal relation between immunological status, tuberculosis infection and smoked tobacco but this association and the occurrence of other infections surely needs further investigation. Limitations We acknowledge three main limitations to our survey. First, as it was conducted in a limited number of HIV/AIDS clinics volunteering and having the capacity to conduct it, these results could not be extrapolated to all the IeDEA participating centers and thus to the entire HIV/AIDS infected population currently in care in West Africa. Another limitation of this work is the lack of reliable data concerning these consumptions in the general population of comparable age and gender distribution so as to compare the impact of such consumptions according to HIV status. Consequently, we cannot actually tell if the relatively high smoking prevalence found in Mali is related to a higher tobacco use in HIV-infected patients or if it is related to the current higher prevalence of smoking in the general population of Mali compared to other countries in the region. Finally, the information related to tuberculosis infection relied on medical records from each participating site. Facing the absence of harmonized definition of tuberculosis and the relative difficulty to assess a diagnosis of certainty, we acknowledge the lack of stability concerning this association. However, facing the great burden of tuberculosis in sub-Saharan Africa and the growing evidence of its association with smoking from previous reports elsewhere in the world, this research question should become a priority. |
This first description of smoking patterns in HIV-infected patients on HAART from three French-speaking countries in West Africa showed marked differences between communities. It highlighted the need for extending this survey to other clinics and regions in this continent in order to better describe these differences and to identify populations highly exposed to this cancer and cardiovascular risk factor. Finally, this survey also provides evidences concerning the association between cigarette smoking and tuberculosis in HIV-infected patients, a major public health issue in this part of the world. |
We are indebted to the interviewers who performed the data collection and to the data clerks who entered all the data in the IeDEA/PAC-CI office in Abidjan. Special thanks to Rosemary McKaig, Caroline Williams, Gerald Sharp and Geraldina Domingez for their support in the design and conduct of these studies.
Source of support
This work was funded by the following institutes: the National Cancer Institute (NCI), the Eunice Kennedy Shriver National Institute of Child Health & Human Development (NICHD) and the National Institute of Allergy and Infectious Diseases (NIAID). |
The IeDEA West Africa Cohort Collaboration is constituted as follows:
-
Principal investigator: Pr François Dabis (INSERM U897, ISPED, Bordeaux, France)
-
Associate investigator: Pr Emmanuel Bissagnene (SMIT, Treichville, Abidjan, Côte d’Ivoire)
-
Co-investigators: Clarisse Amani-Bosse, Franck Olivier Ba-Gomis Emmanuel Bissagnene, Eric Delaporte, Constance Kanga-Koffi, Moussa Maiga, Eugène Messou, Albert Minga, Kevin Peterson, Papa Salif Sow, Hamar Traoré, Marcel D Zannou
-
Other members: Gérard Allou, Xavier Anglaret, Alain Azondékon, Eric Balestre, Jules Bashi, Ye Diarra, Joseph Drabo, Jean-François Etard, Didier K Ekouévi, Antoine Jaquet, Alain Kouakoussui, Valériane Leroy, Charlotte Lewden, Karen Malateste, Lorna Renner, Haby Signaté Sy, Marguerite Timité-Konan, Rodolphe Thiebault, Hapsatou Touré, Annnie J Sasco
-
Coordinating centres
-
1)
HIV Epidemiology, INSERM CRE U 897, ISPED, Université Victor Segalen, Bordeaux, France
-
2)
Epidemiology for Cancer Prevention, INSERM CRE U 897, ISPED, Université Victor Segalen, Bordeaux, France
-
3)
Programme PAC-CI, CHU de Treichville, Abidjan, Côte d’Ivoire
-
Adults Clinical centers
-
4)
Service d’Hépato-Gastro-Entérologie, Hôpital Gabriel Touré, Bamako, Mali
-
5)
Centre de Prise en Charge des Personnes vivant avec le VIH, CHNU, Cotonou, Benin
-
6)
ACONDA-CePReF, Adultes, Abidjan Côte d’Ivoire
-
7)
Centre de Prise en Charge des Personnes vivant avec le VIH, Hôpital du point G, Bamako, Mali
-
8)
Unité de Soins Ambulatoires et de Conseil (USAC), Abidjan, Côte d’Ivoire
-
9)
Centre Intégré de Recherche Bioclinique d’Abidjan (CIRBA), Abidjan, Côte d’Ivoire
-
10)
Centre Médical de Suivi de Donneurs de Sang/CNTS/PRIMO-CI, Abidjan, Côte d’Ivoire
-
11)
Service de Maladies Infectieuses et Tropicales (SMIT) de l’hôpital de Treichville, Abidjan, Côte d’Ivoire
-
12)
ACONDA-MTCT+ Adultes, Abidjan, Côte d’Ivoire
|
1. Lewden C, Salmon D, Morlat P, Bevilacqua S, Jougla E, Bonnet F, et al. Causes of death among human immunodeficiency virus (HIV)-infected adults in the era of potent antiretroviral therapy: emerging role of hepatitis and cancers, persistent role of AIDS. Int J Epidemiol. 2005 Feb;34(1):121–30. 2. Benard A, Tessier JF, Rambeloarisoa J, Bonnet F, Fossoux H, Neau D, et al. HIV infection and tobacco smoking behaviour: prospects for prevention? ANRS CO3 Aquitaine Cohort, 2002. Int J Tuberc Lung Dis. 2006 Apr;10(4):378–83. 3. Collins RL, Kanouse DE, Gifford AL, Senterfitt JW, Schuster MA, McCaffrey DF, et al. Changes in health-promoting behavior following diagnosis with HIV: prevalence and correlates in a national probability sample. Health Psychol. 2001 Sep;20(5):351–60. 4. Friis-Moller N, Weber R, Reiss P, Thiebaut R, Kirk O, d’Arminio Monforte A, et al. Cardiovascular disease risk factors in HIV patients--association with antiretroviral therapy. Results from the DAD study. AIDS. 2003 May 23;17(8):1179–93. 5. Gritz ER, Vidrine DJ, Lazev AB, Amick BC 3rd, Arduino RC. Smoking behavior in a low-income multiethnic HIV/AIDS population. Nicotine Tob Res. 2004 Feb;6(1):71–7. 6. Anonymous. Cigarette smoking among adults--United States, 2006. MMWR Morb Mortal Wkly Rep. 2007 Nov 9;56(44):1157–61. 7. Saves M, Chene G, Ducimetiere P, Leport C, Le Moal G, Amouyel P, et al. Risk factors for coronary heart disease in patients treated for human immunodeficiency virus infection compared with the general population. Clin Infect Dis. 2003 Jul 15;37(2):292–8. 8. Sasco AJ, Merrill RM, Dari I, Benhaim-Luzon V, Carriot F, Cann CI, et al. A case-control study of lung cancer in Casablanca, Morocco. Cancer Causes Control. 2002 Sep;13(7):609–16. 9. Voirin N, Berthiller J, Benhaim-Luzon V, Boniol M, Straif K, Ayoub WB, et al. Risk of lung cancer and past use of cannabis in Tunisia. J Thorac Oncol. 2006 Jul;1(6):577–9. 10. World Health Organization. Guidelines for controlling and monitoring the tobacco epidemic. Geneva: 1998. 11. Shafey O, Eriksen M, Ross H, Mackay J. The Tobacco Atlas. American Cancer Society. World Lung Foundation; 2009 [last accessed 03/10/09]. available at http://www.tobaccoatlas.org/. 12. Koffi N, Kouassi B, Horo K, Kouakou YG, N’Gom A, Toloba Y, et al. Smoking in the African setting (Abidjan, Ivory coast): patient knowledge, attitude and behavior. Rev Pneumol Clin. 2004 Sep;60(4):217–22. 13. Sodjinou R, Agueh V, Fayomi B, Delisle H. Obesity and cardio-metabolic risk factors in urban adults of Benin: relationship with socio-economic status, urbanisation, and lifestyle patterns. BMC Public Health. 2008;8:84. 14. Pampel F. Tobacco use in sub-Sahara Africa: estimates from the demographic health surveys. Soc Sci Med. 2008 Apr;66(8):1772–83. 15. Cherif MH. The tobacco problem in French-speaking Africa and regional perspectives of the French-speaking African tobacco observatory. Promot Educ. 2005;(Suppl 4):45–7. 61. 16. da Costa e Silva VL. Health consequences of the tobacco epidemic in West African French-speaking countries and current tobacco control. Promot Educ. 2005;(Suppl 4):7–12. 54. 17. Legresley E, Lee K, Muggli ME, Patel P, Collin J, Hurt RD. British American Tobacco and the “insidious impact of illicit trade” in cigarettes across Africa. Tob Control. 2008 Oct;17(5):339–46. 18. Townsend L, Flisher AJ, Gilreath T, King G. A systematic literature review of tobacco use among adults 15 years and older in sub-Saharan Africa. Drug Alcohol Depend. 2006 Sep 1;84(1):14–27. 19. Warren CW, Jones NR, Peruga A, Chauvin J, Baptiste JP, Costa de Silva V, et al. Global youth tobacco surveillance, 2000–2007. MMWR Surveill Summ. 2008 Jan 25;57(1):1–28. 20. Simen-Kapeu A, La Ruche G, Kataja V, Yliskoski M, Bergeron C, Horo A, et al. Tobacco smoking and chewing as risk factors for multiple human papillomavirus infections and cervical squamous intraepithelial lesions in two countries (Cote d’Ivoire and Finland) with different tobacco exposure. Cancer Causes Control. 2009 Mar;20(2):163–70. 21. La Ruche G, Ramon R, Mensah-Ado I, Bergeron C, Diomande M, Sylla-Koko F, et al. Squamous intraepithelial lesions of the cervix, invasive cervical carcinoma, and immunosuppression induced by human immunodeficiency virus in Africa. Dyscer-CI Group. Cancer. 1998 Jun 15;82(12):2401–8. 22. Arcavi L, Benowitz NL. Cigarette smoking and infection. Arch Intern Med. 2004 Nov 8;164(20):2206–16. 23. Boschini A, Smacchia C, Di Fine M, Schiesari A, Ballarini P, Arlotti M, et al. Community-acquired pneumonia in a cohort of former injection drug users with and without human immunodeficiency virus infection: incidence, etiologies, and clinical aspects. Clin Infect Dis. 1996 Jul;23(1):107–13. 24. Gordin FM, Roediger MP, Girard PM, Lundgren JD, Miro JM, Palfreeman A, et al. Pneumonia in HIV-infected persons: increased risk with cigarette smoking and treatment interruption. Am J Respir Crit Care Med. 2008 Sep 15;178(6):630–6. 25. Hirschtick RE, Glassroth J, Jordan MC, Wilcosky TC, Wallace JM, Kvale PA, et al. Bacterial pneumonia in persons infected with the human immunodeficiency virus. Pulmonary Complications of HIV Infection Study Group. N Engl J Med. 1995 Sep 28;333(13):845–51. 26. Nagappan V, Kazanjian P. Bacterial infections in adult HIV-infected patients. HIV Clin Trials. 2005 Jul–Aug;6(4):213–28. 27. Sullivan JH, Moore RD, Keruly JC, Chaisson RE. Effect of antiretroviral therapy on the incidence of bacterial pneumonia in patients with advanced HIV infection. Am J Respir Crit Care Med. 2000 Jul;162(1):64–7. 28. Dhamgaye TM. Tobacco smoking and pulmonary tuberculosis: a case-control study. J Indian Med Assoc. 2008 Apr;106(4):216–9. 29. Hassmiller KM. The association between smoking and tuberculosis. Salud Publica Mex. 2006;48( Suppl 1):S201–16. 30. Kolappan C, Gopi PG. Tobacco smoking and pulmonary tuberculosis. Thorax. 2002 Nov;57(11):964–6. |
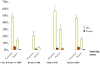 | Figure 1 Estimated smoking prevalence (with 95% upper limit of confidence interval) according to gender and country (n=2920). |
 | Table 1 Main characteristics of HIV-infected adult patients declaring present or former regular tobacco smoking* according to country. IeDEA West Africa collaboration, 2007–2008 |
 | Table 2 Factors associated with tobacco smoking in HIV-infected patients on antiretroviral therapy. IeDEA West Africa collaboration, 2007–2008 |
|

